Why Carbomers Fail the EU’s Microplastic Test (And What It Means)
A Technical Guide to European Commission Regulation 2023/2055 for Beauty R&D leaders
Welcome to Part 2 of our series on the beauty industry’s transition from synthetic to natural high performance polymers. If you missed Part 1 - our love letter to carbomers and why we’re sad to see them go - check it out here.
This week we’re diving into EU regulations on synthetic polymers. It’s a bit dry and technical, but you’ll want to know the ins and outs of this before you start new product development. So go grab another coffee and let’s get started!
The Basics
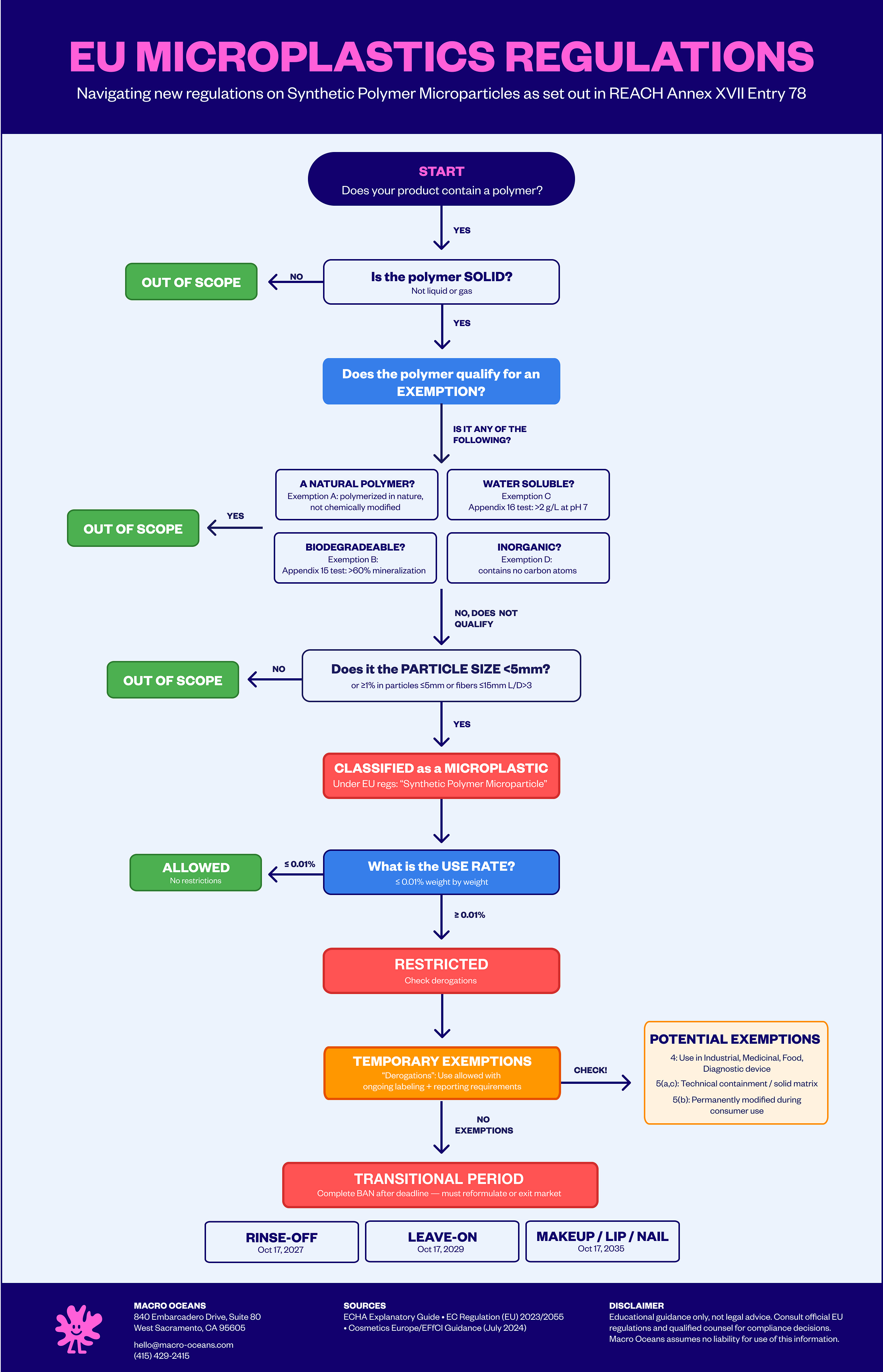
In late 2023, the European Commission published Commission Regulation 2023/2055, restricting the use of “microplastics intentionally added to products”. These rules are formally an amendment to Annex XVII of REACH (the foundational EU legislation designed to protect human health and the environment from chemical risks).
Critically, these EU regulations use the “precautionary principle” which shifts the burden of proof onto the manufacturers who have to demonstrate the chemicals do no harm before the substance is sold. This stance is markedly different from the US, where the onus is on the government to prove the product causes harm after the product is already in the market.
Regulation 2023/2055 specifically restricts the use of “synthetic polymer microparticles”. These are broadly defined as:
Solid polymers that are synthetic or chemically modified
In particles ≤5mm (or length ≤15mm with length-to-diameter ratio >3)
Present at ≥0.01% concentration to confer a “sought-after characteristic”
This definition means not just bans on microbeads and glitter, but also many of the functional synthetic polymers workhorses used for thickeners, stabilizers, film formers and suspensions in formulas.
Are Carbomers Actually Covered?
Given that carbomers disperse in water, it’s fair to ask: are they actually even covered by these regulations? Now for the fun part. A compound is exempt from the regulation if it passes one of the following four tests:
Natural: “polymers that are the result of a polymerisation process that has taken place in nature, independently of the process through which they have been extracted, which are not chemically modified substances”
Solubility: “polymers that have a solubility >2g/L as proved in accordance with Appendix 16”
Degradation: “polymers that are degradable as proved in accordance with Appendix 15”
Inorganic: “polymers that do not contain carbon atoms in their chemical structure”
Carbomers are made up of long chains of cross-linked acrylic acid units. When formulating, a chemist will add them to water and then bring the pH up to neutral. This neutralization step deprotonates the carboxylic acid group and causes the chains to repel each other, creating a matrix to trap the water. The result: they swell dramatically creating beautiful gels.
According to Appendix 16, a compound is soluble when it dissolves into solution at the molecular level at 20°C, pH 7, 10 g/L loading, over 24 hours. Carbomers fail this test. Their crosslinked structures remain intact. Being water swellable does not make them water soluble.
What about degradation? Here carbomers also fail. The criteria set out in Appendix 15 provide mineralization thresholds depending on timeframe and the testing environment. This ranges from >60% mineralization for rapid screens with up to >90% for long-term degradation studies.
At this point, studies have shown that carbomers are non-biodegradable - they are not broken down or metabolized, demonstrating biological persistence. Again, the cross-linked networks mean that their structural chemistry is resistant to biological degradation.
Bottom line: Carbomers are classified as “synthetic polymer microplastics” under this regulation. There are no exemptions that allow their continued use in consumer beauty after the transitional periods expire.
Once you absorb that, the next question is, when do all the changes happen?
The Three Compliance Cliffs
The restriction formally entered into force on October 17, 2023. But the regulations provided transition periods for different types of products:
October 16, 2027: Rinse-off products such as cleansers, shampoos, conditioners, body washes, scrubs, masks
October 16, 2029: Leave-on products such as moisturizers, serums, sunscreens (non-spray), lotions, treatments, eye creams
October 16, 2035: Color cosmetics such as make-up, lip, and nail products. These have the longest runway, but there’s a catch: from October 17, 2031, these products must carry the label “this product contains microplastics” to continue being sold.

For multinational brands with products across all categories, this creates cascading pressure with more than just hero products needing reformulation.
There are a few cases where the regulations allow for temporary exemptions (or “derogations” in the official jargon):
Derogation 4: where the compound is used in industrial, medical, food, or diagnostic device applications
Derogation 5: where the compound is either demonstrated to have been permanently modified during consumer use, technically contained so that it is not released into the natural environment, or permanently incorporated into a solid matrix
For each of these exemptions there are annual reporting requirements to ECHA, labelling requirements to ensure proper use and disposal.
Please make it simple!
Still with me? Or totally confused? Fear not! We’ve created a flow diagram so you can figure out if your beloved polymer of choice is allowed or not.
Download the pdf version here!
I told you this wasn’t straightforward! Clearly, this isn’t “business as usual with a bit of extra paperwork.” Not knowing which polymers are allowed or banned means ongoing regulatory exposure, annual reporting obligations, and—for color cosmetics—four years of mandatory “contains microplastics” labeling that no brand wants on their packaging.
Why This Matters for US Brands
If you’ve made it this far (well done!) you may be thinking: this is an EU regulation and I run a US brand, so why should I care? There are several reasons to consider these new rules.
Future proof your formulations: If you’re developing new products or already selling in both the US and the EU, then reformulating once for global compliance is more cost-effective than managing dual formulations, inventory, and supply chains.
Regulatory anticipation: The EU restriction previews where US regulation could head next. California and New York have already banned microbeads and there is new microplastic legislation under discussion.
Consumer perception: US consumers read the same health headlines about microplastics in human blood and brain tissue as European consumers.
Retail buyer pressure: Most US retailers now have some version of a “restricted substance list” and as microplastics awareness grows, synthetic polymers will likely be added.
What This Means for Your Decisions
The new EU rules are not ambiguous: carbomers and acrylates are covered, the deadlines are set, and the compliance burden for continued use is significant. So bottom line, reformulation is the most obvious answer.
If regulations are creating a forcing function, the next question is: what do consumers really think of microplastics and natural polymers? Find out next week in Part 3 of our series.